| IWRS randomisation | The 'IWRS randomisation' module is built into eSOCDAT – no interface to another external system is required so sites randomise patients directly in eSOCDAT. The module supports open or double-blind, multi-strata, multi-group randomisation based on a block list or a widely configurable range of algorithms such as minimisation. The randomisation list can include blocks of any size linked to an unlimited number of defined patient group criteria and stratification parameters. |
| Trial Supply Management (Drug management) | The 'Trial Supply Management' module enables the capture of all information required to track study treatment supplies which includes information concerning the country / regional depots if used to ship study treatment supplies to the sites and the inventory at each depot. For multinational studies, it is possible to track batch release dates by country – i.e. date when the Qualified Person (QP) release was obtained for each of the participating countries for the batch number concerned. In addition, site (re)supplies can be issued directly from this module either automatically and/or manually – there is a complete tracking from date the request was issued to when received at site. At the site level, the System tracks expiry dates and takes this into consideration when allocating study treatment to any one patient. This module is IRT certified by some of the leading drug shipping companies. |
| Site Monitoring | The 'Site Monitoring' module enables authorised users to register and manage all site contacts (e.g. pre-initiation, initiation, monitoring, close-out visits/email/telephone reports or remote monitoring activities) and to complete the site contact reports directly in eSOCDAT. The content of each visit report can be easily configured to the specific requirements of your study. Site contacts/visit reports can thus be managed and tracked very efficiently in the one system and the tracking of action points can be easily followed until resolution. Sponsors have 24/7 oversight of what’s going on at any time with any site. |
| Risk-based Monitoring | The 'Risk-Based Monitoring' module is an innovative, powerful and scalable tool to enable users to implement the Risk-Based Monitoring (RBM) strategy/rules per the risk-based monitoring plan. This module is extremely flexible as users can implement the RBM rules either by patient, for a subset of patients with a specific demographic (e.g. gender, age) or a specific condition (e.g. diabetes, hypertension) or for specific visit/record types. Users can also define a percentage of patients/visits that require monitoring to be performed. The patients/visits/records requiring monitoring will be displayed in the eSOCDAT Source Data Verification module ('SDV') module. |
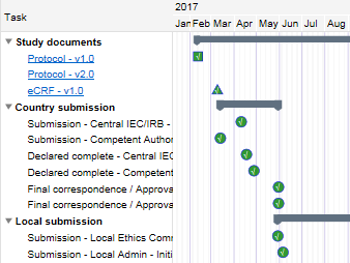
| Study Oversight | The 'Study Oversight' module contains powerful tools enabling all stake holders to have a clear vision and complete, real-time study oversight. Robust metrics and status reports can be made available to authorised users and can be monitored 24/7. Study and site level Dashboards are readily available and easily accessible by users to guide the work flow. Users can track the study milestones including leading or lagging time via the ‘Milestone’ part of this module. Users can create and access metric and status reports which are displayed on the screen to track, amongst others, recruitment progress, key performance indicators and key risk indicators. |
 |
eTMF | The 'eTMF' module enables authorised users to manage (i.e. upload, download, validate, track) documents which are part of the study Trial Master File (TMF). This module is user and role restricted and thus enables authorised users to either upload and/or download documents. The meta data is specific to each document type and place holders can be set-up for documents that require renewal or which are expected to be received. The eSOCDAT eTMF module follows the principles of the Drug Information Association’s (DIA) TMF Reference Model. |
Adjudication | The 'Adjudication' module supports all activities relating to event adjudication. It enables the generation of eCRFs to be completed directly in the System by the site personnel for events assigned for adjudication – each eCRF will be specific to the event type requiring adjudication. The receipt of all source documents required to make up the adjudication packages can be fully tracked thus allowing a real-time status. Assignment of events to adjudicators can be done via a programmed and validated algorithm or performed manually. Adjudicators are notified by email that events are ready for adjudication – the frequency of these mails can be determined for each trial and can be adapted over the life-cycle of the trial. Adjudicators complete the tailored adjudication forms directly in this module. Adjudicators only have access to events assigned to him/her and are not aware of the other adjudicator with whom he/she has been paired to adjudicate the event, thus enabling complete independence between the two persons adjudicating the events. |
Training Tracking | The 'Training Tracking' module enables users to record and track all training performed for site and non-site personnel involved in your study. The training materials/documents used for the training can be easily accessed in the eTMF via a document hyperlink present in the Training Tracking module. Placeholders can be created for training that requires renewal over the life cycle of your study and re-training on a same document version can also be tracked. Access to this module is user and role restricted and each user has access only to his/her training details. Documents which are uploaded in the eTMF and require training to be performed become automatically available for training by specifying the relevant user roles. Each user will (electronically) sign his/her training performed. |
Deviation Management | The 'Deviation Management' module enables users to record and track all deviations which occur over the life-cycle of your study. Patient-related deviations can be created automatically and non-patient related deviations, manually. For each deviation, users can assess the impact of the deviation on patient safety, data quality, and integrity as well as track the action(s) taken for each deviation – e.g. perform a CAPA, or re-train the site personnel etc. From the data compiled in this module, users can easily create a report to send to the Ethics Committee /Institutional Review Board per the local requirements. Sponsors have real-time oversight (24/7) to information concerning the occurrence and management of deviations. |
Submission Tracking | The 'Submission Tracking' module is a powerful tool that enables users to capture and track all information relating to the Regulatory, Ethics/IRB and local administrative submissions. Users have real-time access to the status of each submission type. In addition, via this module, users can compile all documents required for a submission into one file, on the assumption that all documents required for a submission are uploaded in the eTMF. |
Meeting/Payment Tracking | The 'Meeting Tracking' module enables users to track all meetings (e.g. Steering, Adjudication Committee, Sponsor, Internal, Investigator, CRA meetings ) that take place over the life-cycle of a study capturing the meeting date, location and attendees.
The ‘Payment Tracking’ module enables tracking of payments made to sites – e.g. patient related per the milestone payments in the site contract – the milestone payment timings can be tailored per the contract requirements, reimbursements for travel / hotel accommodation costs relating to meetings and/or payments made to Committee members. |
Data Management | The 'Data Management' module enables enables the DM team to efficiently execute all data management/cleaning activities over the life cycle of a study. Tailored data checks are defined and programmed/validated and implemented in the System for each study. The DM team can then run the data checks per the frequency described in the Data Management Plan and post data queries as appropriate for sites to resolve. The DM workflow and progress is guided with powerful and intuitive metric reports generated directly within the System. To facilitate the efficiency of the DM team, when the data checks are run, data queries are automatically inserted into the data query field. Visits can be 'soft-locked' as the data cleaning is performed, thus accelerating the time to database lock at the end of the trial. |
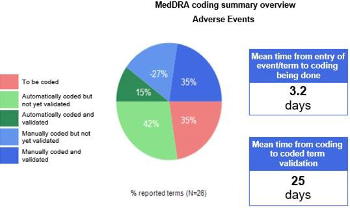
Coding of medical terms and concomitant medications | The 'Coding' module enables the coding of medical free text – e.g. adverse events, free text medical history and concomitant treatments using the Regulatory approved (e.g MedDRA, WHO-DD) respective medical coding dictionaries. The Coding dictionaries are fully integrated in eSOCDAT thus eliminating the need to import/export data requiring coding to another platform. Using powerful search tools, automatic and manual coding of site reported verbatim terms can be done instantaneously after the data has been entered by the sites. The coders can post queries as appropriate for sites directly in the System. Another special feature of this module is the ability for an independent coder to perform a QC check to validate the coding done. Using this module will thus guarantee a timely and accurate coding of verbatim terms and will hence ensure that reliably coded data is always available for safety reporting. |